Abstract
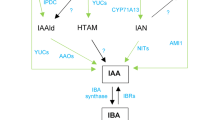
Seaweeds and their extracts have been used for centuries in agriculture to improve plant growth and impart stress tolerance. There has been historical evidence that phytohormones present in seaweeds lead to these effects, but questions of this mode of action have always been raised. By quantifying phytohormones in seaweed extracts coupled with the use of phytohormone biosynthetic and insensitive mutants, we conclude that the phytohormone levels present within the extracts are insufficient to cause significant effects in plants when extracts are applied at recommended rates. However, components within seaweed extracts may modulate innate pathways for the biosynthesis of phytohormones in plants. Phytohormone profiles of plant tissue extracts were analyzed following root application of a commercial seaweed extract produced from Ascophyllum nodosum (ANE) to in vitro-grown Arabidopsis plants. We found an increase in total concentration of cytokinins (CKs), in particular, of trans-zeatin-type CKs, 24 and 96 h after ANE application, with an increase in cis-zeatin-type CKs observed at 144 h. Concomitantly, increases in abscisic acid (ABA) and ABA catabolite levels were observed whereas auxin levels were reduced. Additionally, the profile of transcripts revealed that CK biosynthetic genes were upregulated, whereas the CK catabolic genes were repressed at 24 and 96 h following ANE application. Not surprisingly, the transcripts of ABA biosynthetic genes were increased whereas the auxin biosynthetic genes were repressed. These corroborated findings are the first to help explain the underlying physiological benefits derived from the application of ANE to plants.










Similar content being viewed by others
References
Abrams SR, Nelson K, Ambrose SJ (2003) Deuterated abscisic acid analogs for mass spectrometry and metabolism studies. J Label Compd Radiopharm 46:273–283
Aloni R, Aloni E, Langhans M, Ullrich CI (2006) Role of cytokinin and auxin in shaping root architecture: regulating vascular differentiation, lateral root initiation, root apical dominance and root gravitropism. Ann Bot 97:883–893
Argueso CT, Raines T, Kieber JJ (2010) Cytokinin signaling and transcriptional networks. Curr Opin Plant Biol 13:533–539
Arthur GD, Stirk WA, van Staden J (2003) Effect of a seaweed concentrate on the growth and yield of three varieties of Capsicum annuum. S Afr J Bot 69:207–211
Bartrina I, Otto E, Strnad M, Werner T, Schmulling T (2011) Cytokinin regulates the activity of reproductive meristems, flower organ size, ovule formation, and thus seed yield in Arabidopsis thaliana. Plant Cell 23:69–80
Bishopp A, Benkova E, Helariutta Y (2011) Sending mixed messages: auxin-cytokinin crosstalk in roots. Curr Opin Plant Biol 14:10–16
Blunden G, Jenkins T, Liu YW (1996) Enhanced leaf chlorophyll levels in plants treated with seaweed extract. J Appl Phycol 8:535–543
Boyer GL, Dougherty SS (1988) Identification of abscisic acid in the seaweed Ascophyllum nodosum. Phytochemistry 27:1521–1522
Chiwocha SD, Abrams SR, Ambrose SJ, Cutler AJ, Loewen M, Ross AR, Kermode AR (2003) A method for profiling classes of plant hormones and their metabolites using liquid chromatography-electrospray ionization tandem mass spectrometry: an analysis of hormone regulation of thermodormancy of lettuce (Lactuca sativa L.) seeds. Plant J 35:405–417
Chiwocha SD, Cutler AJ, Abrams SR, Ambrose SJ, Yang J, Ross AR, Kermode AR (2005) The etr1-2 mutation in Arabidopsis thaliana affects the abscisic acid, auxin, cytokinin and gibberellin metabolic pathways during maintenance of seed dormancy, moist-chilling and germination. Plant J 42:35–48
Chomczynski P, Sacchi N (1987) Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Anal Biochem 162:156–159
Craigie J (2011) Seaweed extract stimuli in plant science and agriculture. J Appl Phycol 23:371–393
Crouch IJ, van Staden J (1992) Effect of seaweed concentrate on the establishment and yield of greenhouse tomato plants. J Appl Phycol 4:291–296
Crouch IJ, van Staden J (1993) Evidence for the presence of plant growth regulators in commercial seaweed products. Plant Growth Regul 13:21–29
D’Agostino IB, Deruere J, Kieber JJ (2000) Characterization of the response of the Arabidopsis Response Regulator gene family to cytokinin. Plant Physiol 124:1706–1717
De Smet I, Signora L, Beeckman T, Inze D, Foyer CH, Zhang HM (2003) An abscisic acid-sensitive checkpoint in lateral root development of Arabidopsis. Plant J 33:543–555
De Smet I, Zhang HM, Inze D, Beeckman T (2006) A novel role for abscisic acid emerges from underground. Trends Plant Sci 11:434–439
Fan D, Hodges DM, Zhang JZ, Kirby CW, Ji XH, Locke SJ, Critchley AT, Prithiviraj B (2011) Commercial extract of the brown seaweed Ascophyllum nodosum enhances phenolic antioxidant content of spinach (Spinacia oleracea L.) which protects Caenorhabditis elegans against oxidative and thermal stress. Food Chem 124:195–202
Fornes F, Sãnchez-Perales M, Guardiola JL (2002) Effect of a seaweed extract on the productivity of ‘de Nules’ Clementine Mandarin and Navelina Orange. Bot Mar 45:486–489
Fukaki H, Tasaka M (2009) Hormone interactions during lateral root formation. Plant Mol Biol 69:437–449
Gan SS, Amasino RM (1995) Inhibition of leaf senescence by autoregulated production of cytokinin. Science 270:1986–1988
Ghanem ME, Albacete A, Smigocki AC, Frebort I, Pospisilova H, Martinez-Andujar C, Acosta M, Sanchez-Bravo J, Lutts S, Dodd IC, Perez-Alfocea F (2011) Root-synthesized cytokinins improve shoot growth and fruit yield in salinized tomato (Solanum lycopersicum L.) plants. J Exp Bot 62:125–140
Guo JC, Duan RJ, Hu XW, Li KM, Fu SP (2010) Isopentenyl transferase gene (ipt) downstream transcriptionally fused with gene expression improves the growth of transgenic plants. Transgenic Res 19:197–209
Havlova M, Dobrev PI, Motyka V, Storchova H, Libus J, Dobra J, Malbeck J, Gaudinova A, Vankova R (2008) The role of cytokinins in responses to water deficit in tobacco plants over-expressing trans-zeatin O-glucosyltransferase gene under 35S or SAG12 promoters. Plant, Cell Environ 31:341–353
Khan W, Rayirath UP, Subramanian S, Jithesh MN, Rayorath P, Hodges DM, Critchley AT, Craigie JS, Norrie J, Prithiviraj B (2009) Seaweed extracts as biostimulants of plant growth and development. J Plant Growth Regul 28:386–399
Khan W, Hiltz D, Critchley AT, Prithiviraj B (2011) Bioassay to detect Ascophyllum nodosum; extract-induced cytokinin-like activity in Arabidopsis thaliana. J Appl Phycol 23:409–414
Kieber JJ, Schaller GE (2010) The perception of cytokinin: a story 50 years in the making. Plant Physiol 154:487–492
Kingman AR, Moore J (1982) Isolation, purification and quantitation of several growth-regulating substances in Ascophyllum nodosum. Bot Mar 25:149–153
Kudo T, Kiba T, Sakakibara H (2010) Metabolism and long-distance translocation of cytokinins. J Integr Plant Biol 52:53–60
Kumar G, Sahoo D (2011) Effect of seaweed liquid extract on growth and yield of Triticum aestivum var. Pusa Gold. J Appl Phycol 23:251–255
Kumari R, Kaur I, Bhatnagar A (2011) Effect of aqueous extract of Sargassum johnstonii Setchell on growth, yield and quality of Lycopersicon esculentum Mill. J Appl Phycol 23:623–633
Kuroha T, Tokunaga H, Kojima M, Ueda N, Ishida T, Nagawa S, Fukuda H, Sugimoto K, Sakakibara H (2009) Functional analyses of LONELY GUY cytokinin-activating enzymes reveal the importance of the direct activation pathway in Arabidopsis. Plant Cell 21:3152–3169
Leclerc M, Caldwell CD, Lada RR, Norrie J (2006) Effect of plant growth regulators on propagule formation in Hemerocallis spp. and Hosta spp. HortScience 41:651–653
MacKinnon SL, Hiltz D, Ugarte R, Craft CA (2010) Improved methods of analysis for betaines in Ascophyllum nodosum and its commercial seaweed extracts. J Appl Phycol 22:489–494
Mancuso S, Azzarello E, Mugnai S, Briand X (2006) Marine bioactive substances (IPA extract) improve foliar ion uptake and water stress tolerance in potted Vitis vinifera plants. Adv Hort Sci 20:156–161
Matsumoto-Kitano M, Kusumoto T, Tarkowski P, Kinoshita-Tsujimura K, Vaclavikova K, Miyawaki K, Kakimoto T (2008) Cytokinins are central regulators of cambial activity. Proc Natl Acad Sci USA 105:20027–20031
Merewitz EB, Gianfagna T, Huang BR (2011) Photosynthesis, water use, and root viability under water stress as affected by expression of SAG12-ipt controlling cytokinin synthesis in Agrostis stolonifera. J Exp Bot 62:383–395
Miyawaki K, Matsumoto-Kitano M, Kakimoto T (2004) Expression of cytokinin biosynthetic isopentenyltransferase genes in Arabidopsis: tissue specificity and regulation by auxin, cytokinin, and nitrate. Plant J 37:128–138
Miyawaki K, Tarkowski P, Matsumoto-Kitano M, Kato T, Sato S, Tarkowska D, Tabata S, Sandberg G, Kakimoto T (2006) Roles of Arabidopsis ATP/ADP isopentenyltransferases and tRNA isopentenyltransferases in cytokinin biosynthesis. Proc Natl Acad Sci USA 103:16598–16603
Moubayidin L, Di Mambro R, Sabatini S (2009) Cytokinin-auxin crosstalk. Trends Plant Sci 14:557–562
Nambara E, Marion-Poll A (2005) Abscisic acid biosynthesis and catabolism. Annu Rev Plant Biol 56:165–185
Nibau C, Gibbs DJ, Coates JC (2008) Branching out in new directions: the control of root architecture by lateral root formation. New Phytol 179:595–614
Nooden LD, Kahanak GM, Okatan Y (1979) Prevention of monocarpic senescence in soybeans with auxin and cytokinin - antidote for self-destruction. Science 206:841–843
Okamoto M, Kuwahara A, Seo M, Kushiro T, Asami T, Hirai N, Kamiya Y, Koshiba T, Nambara E (2006) CYP707A1 and CYP707A2, which encode abscisic acid 8′-hydroxylases, are indispensable for proper control of seed dormancy and germination in Arabidopsis. Plant Physiol 141:97–107
Okamoto M, Kushiro T, Jikumaru Y, Abrams SR, Kamiya Y, Seki M, Nambara E (2011) ABA 9′-hydroxylation is catalyzed by CYP707A in Arabidopsis. Phytochemistry 72:717–722
Osmont KS, Sibout R, Hardtke CS (2007) Hidden branches: developments in root system architecture. Annu Rev Plant Biol 57:93–113
Peleg Z, Blumwald E (2011) Hormone balance and abiotic stress tolerance in crop plants. Curr Opin Plant Biol 14:290–295
Perilli S, Moubayidin L, Sabatini S (2010) The molecular basis of cytokinin function. Curr Opin Plant Biol 13:21–26
Pfaffl MW (2001) A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res 29(9):e45
Rayirath P, Jithesh MN, Farid A, Khan W, Palanisamy R, Hankins SD, Critchley AT, Prithiviraj B (2008) Rapid bioassays to evaluate the plant growth promoting activity of Ascophyllum nodosum (L.) Le Jol. using a model plant, Arabidopsis thaliana (L.) Heynh. J Appl Phycol 20:423–429
Rayirath P, Benkel B, Hodges DM, Allan-Wojtas P, MacKinnon S, Critchley AT, Prithiviraj B (2009) Lipophilic components of the brown seaweed, Ascophyllum nodosum, enhance freezing tolerance in Arabidopsis thaliana. Planta 230:135–147
Rivero RM, Kojima M, Gepstein A, Sakakibara H, Mittler R, Gepstein S, Blumwald E (2007) Delayed leaf senescence induces extreme drought tolerance in a flowering plant. Proc Natl Acad Sci USA 104:19631–19636
Rivero RM, Gimeno J, Van Deynze A, Walia H, Blumwald E (2010) Enhanced cytokinin synthesis in tobacco plants expressing P-SARK:iPT prevents the degradation of photosynthetic protein complexes during drought. Plant Cell Physiol 51:1929–1941
Ruzicka K, Simaskova M, Duclercq J, Petrasek J, Zazimalova E, Simon S, Friml J, Van Montagu MCE, Benkova E (2009) Cytokinin regulates root meristem activity via modulation of the polar auxin transport. Proc Natl Acad Sci USA 106:4284–4289
Sakakibara H (2006a) Cytokinin biosynthesis and regulation. In: G.Litwack (ed) Plant hormones. Elsevier, San Diego, pp 271–287
Sakakibara H (2006b) Cytokinins: activity, biosynthesis, and translocation. Annu Rev Plant Biol 57:431–449
Shani E, Ben-Gera H, Shleizer-Burko S, Burko Y, Weiss D, Ori N (2010) Cytokinin regulates compound leaf development in tomato. Plant Cell 22:3206–3217
Sharkey TD, Raschke K (1980) Effects of phaseic acid and dihydrophaseic acid on stomata and the photosynthetic apparatus. Plant Physiol 65:291–297
Shkolnik-Inbar D, Bar-Zvi D (2010) ABI4 mediates abscisic acid and cytokinin inhibition of lateral root formation by reducing polar auxin transport in Arabidopsis. Plant Cell 22:3560–3573
Singh S, Letham DS, Palni LMS (1992) Cytokinin biochemistry in relation to leaf senescence. Endogenous cytokinin levels and exogenous applications of cytokinins in relation to sequential leaf senescence of tobacco. Physiol Plant 86:388–397
Spann TM, Little HA (2011) Applications of a commercial extract of the brown seaweed Ascophyllum nodosum increases drought tolerance in container-grown ‘Hamlin’ Sweet Orange nursery trees. Hort Sci 46:577–582
Stirk WA, van Staden J (1996) Comparison of cytokinin- and auxin-like activity in some commercially used seaweed extracts. J Appl Phycol 8:503–508
Stirk WA, Novák O, Strnad M, van Staden J (2003) Cytokinins in macroalgae. Plant Growth Regul 41:13–24
Stirk W, Arthur G, Lourens A, Novák O, Strnad M, van Staden J (2004) Changes in cytokinin and auxin concentrations in seaweed concentrates when stored at an elevated temperature. J Appl Phycol 16:31–39
Su YH, Liu YB, Zhang XS (2011) Auxin-cytokinin interaction regulates meristem development. Mol Plant 4:616–625
To JPC, Deruere J, Maxwell BB, Morris VF, Hutchison CE, Ferreira FJ, Schaller GE, Kieber JJ (2007) Cytokinin regulates type-A Arabidopsis response regulator activity and protein stability via two-component phosphorelay. Plant Cell 19:3901–3914
Ton J, Davison S, van Wees SCM, van Loon LC, Pieterse CMJ (2001) The Arabidopsis ISR1 locus controlling rhizobacteria-mediated induced systemic resistance is involved in ethylene signaling. Plant Physiol 125:652–661
Wasilewska A, Vlad F, Sirichandra C, Redko Y, Jammes F, Valon C, Frey NFD, Leung J (2008) An update on abscisic acid signaling in plants and more. Mol Plant 1:198–217
Weigel D, Glazebrook J (2002) Arabidopsis: a laboratory manual. Cold Spring Harbor Laboratory Press, Cold Spring Harbor
Werner T, Schmulling T (2009) Cytokinin action in plant development. Curr Opin Plant Biol 12:527–538
Werner T, Motyka V, Strnad M, Schmulling T (2001) Regulation of plant growth by cytokinin. Proc Natl Acad Sci USA 98:10487–10492
Werner T, Motyka V, Laucou V, Smets R, Van Onckelen H, Schmulling T (2003) Cytokinin-deficient transgenic Arabidopsis plants show multiple developmental alterations indicating opposite functions of cytokinins in the regulation of shoot and root meristem activity. Plant Cell 15:2532–2550
Werner T, Kollmer I, Bartrina I, Holst K, Schmulling T (2006) New insights into the biology of cytokinin degradation. Plant Biol 8:371–381
Werner T, Nehnevajova E, Kollmer I, Novak O, Strnad M, Kramer U, Schmulling T (2010) Root-specific reduction of cytokinin causes enhanced root growth, drought tolerance, and leaf mineral enrichment in Arabidopsis and tobacco. Plant Cell 22:3905–3920
Yokoya NS, Stirk WA, van Staden J, Novak O, Tureckova V, Pencik A, Strnad M (2010) Endogenous cytokinins, auxins, and abscisic acid in red algae from Brazil. J Phycol 46:1198–1205
Zaharia LI, Galka MM, Ambrose SJ, Abrams SR (2005) Preparation of deuterated abscisic acid metabolites for use in mass spectrometry and feeding studies. J Label Compd Radiopharm 48:435–445
Zhang XZ, Ervin EH (2004) Cytokinin-containing seaweed and humic acid extracts associated with creeping bentgrass leaf cytokinins and drought resistance. Crop Sci 44:1737–1745
Zhang XZ, Ervin EH (2008) Impact of seaweed extract-based cytokinins and zeatin riboside on creeping bentgrass heat tolerance. Crop Sci 48:364–370
Zhao YD (2010) Auxin biosynthesis and its role in plant development. Annu Rev Plant Biol 61:49–64
Acknowledgments
The research team graciously acknowledges funding received from the National Research Council (NRC), Industrial Research Assistance Program (IRAP), and particularly Dr. D. Douglas without whom this collaborative work would not have been possible. We also thank V. Cekic and M. Lafond (Plant Biotechnology Institute, National Research Council) for hormone-profiling sample preparation and Chaminda deSilva (Nova Scotia Agriculture College) for collection of mutant seeds. BP’s lab is supported by grants from the Natural Sciences and Engineering Research Council of Canada, Nova Scotia Department of Agriculture, and Acadian Seaplants Limited.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
344_2012_9301_MOESM1_ESM.tif
Supplementary Fig. 1: Root growth and development of abi4-1 and ipt1,3,5,7 mutants affected by ANE treatment. A Average length of primary roots in Col-0, abi4-1, and ipt1,3,5,7 seedlings 5 days after transfer to indicated media. The data represent the mean ± SE. B Number of lateral roots per centimeter of newly grown roots at all developmental stages (including lateral root primordial) in seedlings 5 days following transfer
344_2012_9301_MOESM2_ESM.tif
Supplementary Fig. 2: Effect of ANE on root growth of ABA and CK mutants. Representative photos of 12-day-old seedlings of abi4-1, wild-type Col-0, or quadruple ipt1,3,5,7 mutant Arabidopsis 8 days after transfer to control (A) or ANE (B) (0.01 % w/v) and ½ MS solid media
344_2012_9301_MOESM3_ESM.tif
Supplementary Fig. 3: Absolute IAA levels in ANE-treated plants. Endogenous IAA levels from two independent trials. Error bars represent mean ± SE
344_2012_9301_MOESM4_ESM.tif
Supplementary Fig. 4: Expression of the key CK one-step activating genes LOG1,7 and LOG8 at indicated time points following ANE application. Data represent the mean ± SE
344_2012_9301_MOESM5_ESM.tif
Supplementary Fig. 5: Expression of the key GA biosynthetic genes GA2ox2 and GA3ox1 at indicated time points following ANE application. Data represent the mean ± SE
Rights and permissions
About this article
Cite this article
Wally, O.S.D., Critchley, A.T., Hiltz, D. et al. Regulation of Phytohormone Biosynthesis and Accumulation in Arabidopsis Following Treatment with Commercial Extract from the Marine Macroalga Ascophyllum nodosum . J Plant Growth Regul 32, 324–339 (2013). https://doi.org/10.1007/s00344-012-9301-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00344-012-9301-9